The 21st international congress of the European Society of Toxicology In Vitro (ESTIV2022) offers great opportunities to both commercial and non-commercial organizations, allowing them to showcase their products and services.
ESTIV is the leading organization in Europe that strengthens the scientific network of in vitro toxicologists and promotes in vitro toxicology, both scientifically and educationally in all countries of Europe.
Please apply to exhibit and sponsorship and:
- Present your business to an international premier scientific network of in vitro toxicologists
- Get in front and meet new customers – explore new business opportunities
- Promote/market your business/services
Downloads
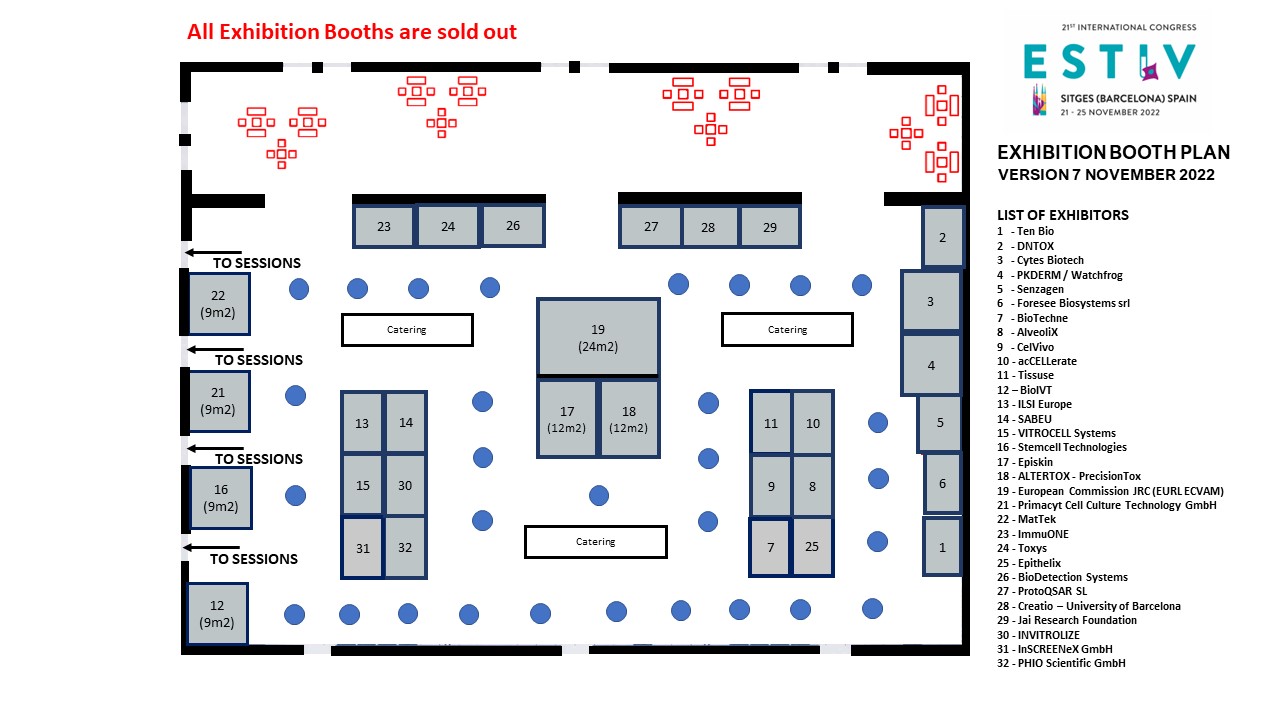
Exhibition Booth Plan
For questions with regard to the exhibition and sponsorships, please contact the Congress Secretariat:
Klinkhamer Group | conferences & events
Tel.: +31 43 362 7008
Email: helpdesk@klinkhamergroup.com
Silver sponsor
Bronze sponsors (in alphabetical order)
Other sponsors and exhibitors (in alphabetical order)

Short profile of the companies
Alternatives Research & Development Foundation (ARDF)
The Alternatives Research & Development Foundation (ARDF) funds innovative research to develop non-animal methods for research, testing, and education. Through grant programs, achievement awards, and sponsorship of scientific conferences, ARDF works constructively with partners in the scientific community to bring alternatives technology and compassion to modern biomedical research, product testing, and classroom laboratories. Established in 1993, ARDF has awarded over $4 million in grants to advance alternative methods that reduce reliance on animal models and promote human-centered science. The Annual Open Grant program is ARDF’s flagship program that supports research across a broad range of scientific areas, from cutting-edge biomedical studies to toxicity and regulatory safety testing. ARDF promotes alternatives through the sponsorship of scientific meetings around the world, participating in regulatory and industry meetings, and providing consultations with media representatives, scientists, and government officials. Visit https://www.ardf-online.org to learn more.
Website: www.ardf-online.org
Company page LinkedIn: ARDF
Contact person via LinkedIn: Angela Hvitved
acCELLerate GmbH
acCELLerate is a specialist for large-scale production and cryopreservation of highly functional assay ready cells. The instant use of cells from a frozen stock became a widely accepted approach preferred to continuously passaged cells. Assay ready cells are used manifold for the screening of new drugs, the animal free safety testing, and for the potency testing of biotherapeutics in clinical development and manufacturing control. Assay ready cells increase the flexibility of any cell-based project and can significantly improve assay precision.
Based on 16 years of experience acCELLerate has developed freezing protocols and established well-controlled processes to prepare assay ready Frozen Instant Cells of high quality and robustness. Frozen Instant Cells can be used like a reagent without prior cultivation. acCELLerate provides assay ready cells from multiple cell lines, validated instaCELL bioassay kits, as well as custom cell banks of Frozen Instant Cells with comprehensive documentation to meet specific requirements of the different fields of application.
Website: www.accellerate.me
Contact person via LinkedIn: Oliver Wehmeier
Altertox – PrecisionTox
Since 2012 Altertox’ purpose has advocated 3Rs to the Members of the European Parliament. Currently Altertox has expanded its activity to education & training, consulting, as well as communication and dissemination of scientific results of EU projects to targeted audiences in particular policy makers and peers using innovative formats and social networks. In 2022, particular attention was given to the general public by developing educational material targeting primary and secondary schools to learn more about 3Rs.
Website: academy.altertox.be
Company page LinkedIn: Altertox
Contact person via LinkedIn: Valentin Salamone
The goal of PrecisionTox is to improve chemical safety assessment to better protect human health and the environment by using non-traditional test species, multiple fields of knowledge, and powerful computational approaches to understand which chemicals are toxic and why.
Website: precisiontox.org
AlveoliX
General information
AlveoliX provides advanced in-vitro models, called Organs-on-Chip. These models are used to better predict human responses in preclinical drug development and to gain more insight into human biology.
AlveoliX’s mission is to make organs-on-chips the new standard for preclinical decision-making and the leading alternative to animal experiments. With our technology, we reproduce the microenvironment of human organs in an unprecedented way.
AXBarrier-on-Chip System
AlveoliX’s Organs-on-Chip Technology allows to reproduce the microenvironment of human organs in an unprecedented way. To make our technology available we developed the AXBarrier-on-Chip System. It enables modelling different organ barriers on chip to simulate the in-vivo conditions of human organ. The possibility of applying 3D stretch to the ultrathin membrane stimulates the cells, keeping them functional and in a physiological shape. Using the AXBarrier-on-Chip System healthy, diseased, and personalized in-vitro models can be created.
The system is easy to handle and compatible with standard lab equipment to minimize the translation time to your lab. It is fully compatible with advanced and conventional cell and molecular biology readouts including barrier function assessment (flux assays and TER). It enables comprehensive molecular analysis on the gene and protein level (e.g., sequencing, cytokine, and metabolic testing) and live-cell or high-resolution imaging.
You will gain a new technology:
• To model more physiologically different barriers like lung, gut, skin, brain, kidney, bladder etc.
• To create mono-, co- or multiple-culture models on-chip
• To create static or dynamic biomodels
How can we work together?
Lab Package
AlveoliX Organs-on-Chip Technology can be implemented directly in your lab with full support to set up the system and the bio-model. This means that after the training and the bio-model setup, you can be independent and fully flexible to perform your own experiments.
CRO services
AlveoliX offers safety and efficacy testing on advanced lung in-vitro systems and organs-on-chips.
We are ready to support you in all steps of the experimental design, data analysis, and reporting.
Website: alveolix.com
Company page LinkedIn: AlveoliX
Contact person via LinkedIn: Léa Todeschini
ASSCT
More information will follow soon.
BioDetection Systems
BDS is specialized in mechanism-based biological monitoring to protect human health and the environment. As such BDS offers an extensive panel of high-throughput-compatible (HTPS) CALUX® reporter assays for testing e.g. endocrine disrupting chemicals according in vitro EATS panel for (anti-) Estrogenic (OECD TG455), (anti-) Androgenic (OECD TG458), (anti-) Thyroid (TG in development) and Steroidogenesis (TG456). This enables hazard identification of critical compounds (e.g., cosmetics, biocides, plastic additives) and their impact in complex mixtures (e.g., plastic migration, water or formulations). The assay panel can be used to rapidly evaluate major types of toxicity relevant for regulatory risk assessment, allowing assurance of consumer safety and -confidence. Quantitative measurements allow assessment of points-of -departure in chemical risk assessment, replacing animal experimentation. Specific work-up methods coupled to quantitative measurements allow precise mixture effect assessment of specific chemical classes. Methods have been extensively and independently validated and are included in relevant international guidelines (also ISO 19040-3 and ISO 24295 for ER and AhR CALUX). Since our assays target key pathways in cells and human physiology (such a cytotoxicity, genotoxicity p53, oxidative stress Nrf2, obesity PPAR and early warning PXR), we increasingly employ these methods in the discovery of compounds and compound mixtures with beneficial health effects. BDS provides these services through its own service laboratory, and under training and license for partner laboratories across the globe.
The BDS service laboratory offers a variety of biological and chemical analytical techniques for safety and bioactivity assessment in a wide range of areas, according to standardized operational procedures. In many cases the analysis reports already fulfil the clients’ needs. For more complex projects our experienced staff, which comprises (molecular) biologists, chemists, and toxicologists, may be involved in data interpretation, consultancy and project management. In addition to that, our research experts and facilities are available for contract research and validation studies (e.g., EU projects and tenders).
Website: biodetectionsystems.com
Contact person via LinkedIn: Peter Behnisch
BioIVT
BioIVT is a leading global provider of research models and value-added research services for drug discovery and development. We specialize in control and disease-state biospecimens including human and animal tissues, cell products, blood and other biofluids.
Website: bioivt.com
Company page LinkedIn: BioIVT
Bio-Techne®
Bio-Techne® unites some of the most referenced brands in Life Sciences: R&D Systems®, Novus Biologicals®, Tocris Bioscience ®, ProteinSimple® and Advanced Cell Diagnostics®. Our family of brands collaborate to provide over 350,000 products for Life Science research including recombinant proteins, antibodies, immunoassays, and custom services. We have over 300,000 journal citations, showing that our combined experience is trusted by researchers. We are passionate about scientific discovery, which drives us to manufacture high-quality reagents and award-winning tools for the Life Science and Clinical Diagnostic markets. We understand our customers and we really care about their research. To ensure researcher’s experiments are successful, we endeavour to provide reliable high-performance innovative solutions that they require for scientific discovery and progress.
Website: www.bio-techne.com
Company page LinkedIn: Bio-Techne®
Contact persons via LinkedIn: Yas Heidari
CelVivo
CelVivo provides a 3D cell culture technology based on the clinostat principle, hence its name The ClinoStar System. The dynamic culture environment ensures low shear-stress and diminished depletion zones allowing the cells of choice to assemble into 3D constructs. The 3D structures such as spheroids, organoids or tumoroids maintain or regain in vivo functionality resembling the tissue of origin and can be grown over long time. Establishing a 3D model system is highly relevant when studying toxicology, both acute and accumulated.
Website: celvivo.com
Contact person via LinkedIn: Thomas Sode Faurschou
Cosmetics Europe
Innovitox is an independent scientific consultancy & service provider to industry, academia and non-profit organizations active in the field of cell-based technologies for R&D and safety testing. Its services range from association management and support, project management, meeting organization and scientific writing.
Contact person via LinkedIn: Erwin van Vliet
CREATIO
AVANTDRUG
Creatio is the production and validation center of advanced therapies of the University of Barcelona. Creatio has recently developed a new neurotoxicology and drug screening platform, Avantdrug. Avantdrug is focused on neuronal models obtained from human pluripotent stem cells for in vitro tests. We also offer in vivo services with animal models for neurodegenerative disorders. Avantdrug can develop projects and preclinical regulatory tests under ISO9001, GLP or GMP quality standards.
Creatio, through Avantdrug, offers developmental neurobiology assays, including neurite outgrowth, network evaluation and mature functionality by using high throughput and high cell content analyses.
Avantdrug focuses drug screening approaches on in vitro human models for neurodegenerative disorders as well as on drug screening: neurodegenerative models, neural survival and maturation, and neuroinflammation analysis.
Creatio’s team has more than 25 years of experience working on neurodegenerative disorders and we can provide scientific and technical advice and training.
Website: CREATIO
Contact person via LinkedIn: Dr Josep M. Canals Coll
Cytes Biotechnologies
Cytes Biotechnologies is a biotechnological company that offers products and services (CRO) based on tissue procurement, cell isolation and cell solutions for in vitro models addressed to the research community worldwide.
The company’s main focus is the isolation and full characterization of primary human and animal hepatic and skin cells, as well as providing different tissue explants (pathological and healthy donors).
Cytes Biotechnologies offers an extensive portfolio of fresh and cryopreserved human and animal cells. Among others, plateable and suspension hepatocytes, stellates, Kupffers, hepatic endothelial, liver sinusoidal endothelial cells, liver subcellular fractions, skin fibroblasts, and keratinocytes.
Website: www.cytesbiotechnologies.com
Company page LinkedIn: Cytes Biotechnologies
Contact persons via LinkedIn:
Directorate of Animal Rights Spain
More information will follow soon.
DNTOX
DNTOX offers a bioanalytical testing system for animal-free screening of chemicals regarding their potential to disturb human brain development and cause developmental neurotoxicity (DNT). Our in vitro testing system uses human cell models and enables artificial intelligence-based chemical screening with in-house developed software. Current procedures for regulatory risk assessment of chemicals and drugs regarding their potential to cause developmental neurotoxicity are time-consuming, expensive, and ethically controversial due to the use of laboratory animals. Because of species differences, they have limited predictivity for humans. Our testing strategy is faster, less expensive, animal-free, and the results of the testing have higher relevance to humans. We want to realize safe chemicals without animal testing. The DNTOX test system is a human cell-based testing system developed at the IUF – Leibniz Research Institute for Environmental Medicine in Düsseldorf, which in its entirety models necessary key processes of human brain development in vitro. The animal free testing system identifies substances disturbing neurodevelopmental processes and causing DNT cost- and time-efficiently, reliably, with higher relevance for humans.
Website: dntox.de
Company page LinkedIn: DNTOX
Contact persons via LinkedIn: Silke Beaucamp
Episkin
More information will follow soon.
Epithelix
Established since March 2006 in Plan-Les-Ouates, district of Geneva, as a biotech company, Epithelix Sàrl is specialized in tissue engineering. We provides in vitro alternatives to animal testing (NAMs) to evaluate the effect of new drugs and to perform toxicity testing of chemicals.
Epithelix has developed a unique technology for long-term culture of fully primary in vitro human reconstituted tissues. The company has focused on the human respiratory tract and launched in 2006 upper airway models (MucilAir™), in 2016 lower airway models (SmallAir™) and intent to launch in 2023 an alveolar Model (AlveolAir™).
These models can be provided as a ready-to-use product to any laboratory worldwide or we can contracted as a CRO to perform testing services on our models, on a wide range of application.
Website: www.epithelix.com
Company page LinkedIn: Epithelix
European Commission JRC (EURL ECVAM)
EURL ECVAM is an integral part of the Joint Research Centre (JRC), the science and knowledge service of the European Commission. We are located on the JRC site in Ispra, Italy.
EURL ECVAM promotes and facilitates the use of non-animal methods in testing and research.
For over 30 years, the JRC has been working on the Three Rs – the Replacement, Reduction and Refinement of animal experiments.
The mandate of EURL ECVAM set out in EU legislation on the protection of animals used for scientific purposes and includes a number of duties:
- Research and development, supported through collaboration with EU and international research initiatives
- Validation of alternative methods for the safety assessment of chemicals
- Dissemination of information and sharing of knowledge on the Three Rs
- Promotion of the Three Rs and non-animal approaches in science
Website links:
- EU Reference Laboratory for alternatives to animal testing (EURL ECVAM)
- EU Science Hub – science and knowledge for Europe
Company page LinkedIn: EU Science, Research and Innovation
Company page Twitter: EU Science, Research and Innovation
> #ECVAM
> @EU_ScienceHub
Foresee Biosystems SRL
Foresee biosystems SRL, an italian start-up born in 2021, aims at designing and marketing innovative platforms for in-vitro cardiac electrophysiology and the prediction of toxicological outcomes of a drug or chemical on cellular models. Combining high quality data from robust in vitro Micro Electrode Array (MEA) assays and a unique LASER technology, we enable the recording of intracellular action potentials, adding value and relevance to the toxicity data supplied to our customers in the field of basic research and pharmaceutical industries. The core technology is a patented, non-invasive method for the recording of cell’s action potentials by means of cell poration with a laser-based technique. Our method exploits a laser system to open nanopores in the cardiac cells’ membrane, thus allowing for the precise recording of intracellular action potentials and the short term (or acute) detection of drug-related adverse reactions on the cell’s membrane ion channels. Furthermore, the extremely low-invasiveness of our technology allows, for the first time, the long-term (or chronic) assessment of in-vitro cardiac toxicity, providing the most precise electrophysiological assay on human-derived cardiac cells available on market with the goal to reduce development costs for drugs and dramatically increase safety. In 2022 we have been awarded by the European commission with SiMulTox, a project that aims to develop the first multiparametric platform for functional and structural chronic cardiotoxicity assessment.
Website links:
Company page LinkedIn: Foresee Biosystems SRL
Contact persons via LinkedIn:
Humane Society of the United States
Research and Toxicology Department
Together, the Humane Society family of organizations is the largest force for animal protection globally, active on the ground in over 50 countries across the Americas, Europe, Asia & Africa, protecting animals in the areas of agriculture, fashion, food, science, wildlife, among others.
The HSI Research & Toxicology team includes professionals in the areas of science, policy and public affairs. We work around the globe in cooperation with researchers, companies, governments and like-minded stakeholders to shift the paradigm in toxicology and health research to make human biology the gold standard, which can best be studied using a suite of modern non-animal technologies, together with an understanding of human disease/adverse outcome pathways. Our team works through intergovernmental bodies to accelerate global adoption of modern non-animal methods (NAMs), and through our network of offices to have these approaches taken up through national regulations.
We spearhead the Animal-Free Safety Assessment (AFSA) Collaboration, which brings together corporate and non-profit leaders who share the goal of accelerating a modern, species-relevant approach to safety assessment and hasten the replacement of animal testing. The AFSA Collaboration currently focuses in three areas: industrial chemicals, cosmetics, and biologicals.
We also coordinate the Biomedical Research for the 21st Century (BioMed21) Collaboration, which brings together leading scientists and institutions from across the globe to advance a shared vision of human-focused approach to medical research. Together, we address barriers to progress, opportunities for change and priorities for future research funding.
By taking a solutions-based approach, we are laying the foundation for a safer and healthier global future without the need for animal experimentation.
Website links:
Company page LinkedIn: Humane Society of the United States
Contact person via LinkedIn: Troy Seidle
ILSI Europe
“Collaborative science for safe, nutritious and sustainable food” is what ILSI Europe stands for.
ILSI Europe is the European Branch of the International Life Sciences Institute (ILSI), a global, nonprofit federation. Our mission is to convene the best scientists from academia, industry & public sector to deliver, communicate and disseminate science-based solutions. These solutions ensure foods are safe, nutritious and sustainable, and that they improve planetary and human health and well-being in the 21st century. Our fit-for-purpose deliverables of highest quality and integrity are peer-reviewed publications, Black & White guiding document, workshops, symposia and many more.
Website: ilsi.eu
Company page LinkedIn: ILSI Europe
Contact person via LinkedIn: Isabelle Guelinckx
ImmuONE
ImmuONE is a specialist CRO for in vitro inhalation toxicity assessment providing tailored assessment strategies, new alternative in vitro models and animalproduct-free approaches to inhalation safety.
Tailored assessment strategies: We use a breadth of commercial and non-commercial in vitro cell/tissue models spanning from the nasal to alveolar regions. As well as offering standard endpoints for in vitro toxicity (e.g. viability), we work alongside our clients to provide a range of relevant biochemical, morphological and functional assessments for toxicology and pharmacology to meet discovery and early product development needs. In addition to well-establishedepithelial endpoints, we are experts in the immunological responses of the airways in vitro and provide reliable and reproducible assessment packages for alveolar macrophages to support a more detailed mechanistic understanding of the safety and fate of inhaled substances in the lung.
New in vitro models: We have developed our own in-house immunocompetent cell culture platforms of the alveoli for next generation inhalation risk assessment. ImmuPHAGETM is a robust and reproducible model of human alveolar-like macrophages which closely resembles in vivo morphology, biochemistry and functionality. Our patented system, ImmuLUNGTM is our human alveolar tissue model comprising alveolar epithelial cells and alveolar macrophages in co-culture, and able to provide a detailed understanding of immune and barrier responses in the airways.
Animal product-free approaches: We are committed to reducing the quantity of animal-derived materials and enhancing the human relevance of in vitro cell culture assessment. We have established in-house animal product-free cell culture methodologies and assessment strategies which are at least equivalent to their animal product-containing counterpart and support clients in making their inhalation toxicity assessments more human relevant.”
Website: immuone.com
Company page LinkedIn: ImmuONE
inSCREENex
The core business of InSCREENeX is genetic engineering of mammalian cells. InSCREENeX offers two complementary technologies:
- Generation of stable recombinant cell lines expressing the desired gene(s) of interest (SCREENflex),
SCREENflex establishes your desired cell test system (e.g., GPCRs, ion channels, antibodies, cytokines) within a rapid timeframe and with unmatched expression characteristics. Importantly, different transgenes of interest can be expressed and if desired also with different expression levels.
- Functional immortalization of primary cells (CI-SCREEN)
CI-SCREEN enables the generation of physiologically relevant cell lines from any mammalian primary cell type. CI-SCREEN is based on a gene library, which identifies the optimal cell expansion regimen for the desired cell type. Until now, ISX used CI-SCREEN to establish novel cell systems from 12 different species, from more than 25 cell types and from more than 100 donors.
Cell types available off-the-shelf are
- Endothelial cells
- Osteoblasts
- Thyrocytes
- Bronchial epithelial cells
- Alveolar epithelial cells
- Astrocytes
In addition, InSCREENeX offers the generation of
- personalized cell systems from healthy and diseased individuals,
the generation of 3D in vitro test systems (e.g. spheroids, organoids, in vitro differentiated 3D transwell constructs) employing the functionally immortalized CI-SCREEN cell lines
Website links:
Company page LinkedIn: inSCREENex
Contact persons via LinkedIn: Tobias May
Invitrolize
Invitrolize is a spin-off from the Luxembourg Institute of Science and Technology. We offer innovative 3D alveolar in vitro models for the prediction of chemical respiratory sensitizers and irritants. These in vitro models allow for the first time the in vitro prediction of respiratory sensitization under realistic exposure conditions. The model has been submitted to ECVAM for pre-validation. Our company and team come from the scientific & research sector, and we will continue to maintain the same high quality standards in everything we do. Our products deliver the most reliable results because we only focus on one thing, data. We want to lead the market in alveolar in vitro models for toxicological analysis and drug testing. To this end, we have the support of a network of research centres that allows us to continue carrying out research activities and improve our platform.
Website: www.invitrolize.com
Company page LinkedIn: Invitrolize
Contact persons via LinkedIn: Arno Gutleb
IIVS
IIVS is a GLP-compliant Contract Research Organization focused on non-animal alternatives to toxicology and product safety testing. We assist companies and other institutions in the implementation of in vitro testing strategies that limit animal use. In addition, IIVS assists in the validation of New Approach Methodologies (NAMs) to traditional assays. Our services include assessments of: dermal and ocular irritation and corrosion, dermal sensitization, photosafety, genetic and respiratory toxicology. As a non-profit entity, our laboratory activities and contributions fund our Education and Outreach programs, providing extensive training and active involvement in the advocacy and promotion of non-animal methods worldwide.
Website: www.iivs.org
LinkedIn page organization: IIVS
Contact person via LinkedIn: Brent Gilbert
Jai Research Foundation
Jai Research Foundation (JRF Global) is a contract research organization that offers laboratory studies under GLP to support development of pharmaceutical, veterinary, cosmetic, chemicals, pesticides and biocidal products. Our lab supports studies related to physico-chemical properties, analytical chemistry, toxicity and ecotoxicity.
JRF is fully committed to the principles of the 3Rs, – Replacement, Reduction and Refinement – of animal testing. In accordance with the new legislation requirements (i.e. Registration, Evaluation, Authorization and restriction of chemical regulation or cosmetics Regulation), JRF has developed alternative studies including in vitro ADME, OECD-based in vitro studies, non-GLP cell-based studies and other NAM’s including in silico and read-across.
Website: www.jrfglobal.com
LinkedIn page organization: Jai Research Foundation
JSAAE
After the establishment in 1989, the Japanese Society for Alternatives to Animal Experiments (JSAAE) has been promoting 3Rs research and their social implementation in Japan through a wide variety of activities. We have been contributing many OECD TGs with JaCVAM (Japanese Center for the Validation of Alternative Methods) and had a 6th World Congress in Tokyo in 2007. In terms of applications of new technologies, we have been involved in national projects such as AI-based hazard prediction (2017-2021) and MPS for pharmaceuticals developments (1st phase, 2017-2021; 2nd phase, 2022-2026). Simultaneously, we have been developing international cooperation with EUSAAT, ESTIV and ASCCT as well as Asian counterparts. The next step is to establish Asian Federation of Societies for Alternatives to Animal Experiment. Toward this, we first held the Asian Congress series in Karatsu, Japan, in 2016, which was continued by Chinese Society in Guangzhou, China in 2018 as the second one, and then the 3rd one (3rd ACAAE) will be hosted by KSAAE (Korean Society for Alternatives to Animal Experiments) in this year, December 14-16, 2022 in Cheju Island in Korea. Primary mission of Asian Federation is to support this series of regional conferences. JSAAE, KSAAE, SAAE-I (Society for Alternatives to Animal Experiments-India) and TATT (Society of Toxicological Alternatives and Translational Toxicology, Chinese Society of Toxicology) wish to have such Federation, as well as other new Asian societies such as SAAT-SL (Society for Alternatives to Animal Testing in Sri Lanka) do. We strongly hope we can have a signing ceremony in coming 3rd Asian congress in this December in Korea for establishment of the Asian Federation.
Website: www.asas.or.jp/jsaae
LUSH Prize
The Lush Prize was launched in 2012 to rewards initiatives across science, policy and campaigning which work to end animal testing and achieve a transition towards human-relevant science and the use of new approach methods, particularly in the area of toxicity testing. There are five award categories: Lobbying, Training, Public Awareness, Science, and Young Researchers. Since launch the Prize has awarded 3 million EUR across over 100 projects. The Lush Prize is pleased to support ESTIV 2022.
Website: www.lushprize.org
Contact person via LinkedIn: Rebecca Ram
MatTek Life Sciences
MatTek Life Sciences is at the forefront of tissue engineering technology and its lab-grown human tissues of the skin, eye, respiratory, and digestive systems are used worldwide as ethical and human-relevant alternatives to animal testing. MatTek tissues have unparalleled reproducibility and multiple regulatory acceptances and validations from organizations like the OECD, ISO, and US EPA to predict the irritation, corrosion, and photosensitivity potential of chemical, pharmaceutical, cosmetic, and other skin care products.
Our EpiDerm and EpiOcular models are validated by OECD as an alternative to animal testing, Skin irritation test OECD TG 439, skin corrosion test OECD TG 431, phototoxicity OECD TG 498, eye irritation test OECD TG 492, and biological evaluation of medical devices, ISO 10993-23.
At the present time, MatTek IVLSL produces 8 models including EpiDerm, EpiDerm FT, EpiOcular, EpiIntestinal, EpiIntestinal FT, EpiAirway, EpiOral, and MelanoDerm. MatTek tissues are shipped weekly from its locations in the US and Europe via standard courier services and delivered ready to use with comprehensive testing results obtained in as little as 24 hours. With a footprint, a fraction of the size of animal testing, MatTek’s tissue technology offers a sustainable, streamlined testing option with reduced costs and timelines.
In fulfillment of its mission to expand the accessibility of lab-grown human tissues, MatTek continues to grow its product offerings at all locations and enhance the complexity of existing organ models through robust R&D. MatTek also supports researchers adopting the technology with comprehensive training and the highest-rated technical support in the industry. By providing a platform to obtain human-relevant data that is predictive of clinical outcomes, MatTek is helping researchers achieve their goals of producing reliable products and therapies for consumers and patients.
Website: www.mattek.com
Contact persons via LinkedIn:
Merck Healthcare
Merck, a leading science and technology company, operates across healthcare, life science and electronics. Around 60,000 employees work to make a positive difference to millions of people’s lives every day by creating more joyful and sustainable ways to live. From advancing gene editing technologies and discovering unique ways to treat the most challenging diseases to enabling the intelligence of devices – the company is everywhere. In 2021, Merck generated sales of € 19.7 billion in 66 countries.
Scientific exploration and responsible entrepreneurship have been key to Merck’s technological and scientific advances. This is how Merck has thrived since its founding in 1668. The founding family remains the majority owner of the publicly listed company. Merck holds the global rights to the Merck name and brand. The only exceptions are the United States and Canada, where the business sectors of Merck operate as EMD Serono in healthcare, MilliporeSigma in life science, and EMD Electronics.
Website: www.merckgroup.com
Company page LinkedIn: Merck Healthcare
PHIO Scientific GmbH
PHIO scientific GmbH is the partner of choice for cutting-edge, automatic cell monitoring solutions for research and industry applications.
Our mission – “Life sciences made easy”. We want to improve the quality and data output of cell culture experiments, to work towards a healthier and smarter world. Our technologies streamline the processes through which data-based insights are gained and knowledge is created.
Use our fully automatic, super compact microscopes and cutting-edge AI analysis platform. Benefit from continuous imaging inside the incubator and track cell growth, viability, motility and phenotype – on the fly.
Website: www.phio.de
Contact persons via LinkedIn: Dr. Anna Jötten
PKDerm
PKDERM is a French independent company that provides smart innovative in vitro solutions for safety and efficacy testing.
Created in 2018, PKDERM relies on the expertise of more than 20 years of its founder, Dr. Hanan Osman-Ponchet. As a former leader in drug development for large pharmaceutical companies, PKDERM’s CEO has developed and tested many alternatives to animal experiment models allowing a reliable and secure evolution from in vivo experimentation to in vitro experimentation.
The company is located in Grasse, in the south of France and aims to support partners in the area of cosmetic/personal care, dermatology, pharmaceutical, chemical, nutraceutical, and medical device industries.
At PKDERM, we are familiar with an array of different assay systems to evaluate the safety and efficacy of new products. This allows us to work with our clients to determine the optimum assay that best fit their needs.
Our acknowledged experience with our worldwide network of multidisciplinary partners enables us to rapidly support our customers confronted with new urgent testing needs.
Our biological models include but not limited to: 2D culture of human skin cells (keratinocytes, fibroblasts, melanocytes), 3D human skin equivalent models, excised human skin, 2D and 3D human hepatocytes and 2D and 3D human lung models.
Our in vitro safety testing includes but not limited to: dermal absorption, skin and eye irritation, skin and lung sensitization, cytotoxicity and phototoxicity, endocrine properties, drug metabolism and PK, drug transporters and drug-drug interactions.
Our in vitro pharmacology/efficacy testing includes but not limited to: dermal and nail absorption, inflammation, pigmentation, ageing, wound healing and fibrosis.
We provide personalized service for every client and every project, and we support the development of new products from the initial reflection and conceptualization to the data promotion and publication. We perform standard testing services as well as optimizing and validating new alternative assays.
PKDERM also offers scientific and project management services related to alternative methods to animal testing and provides workshops and practical training on the use of in vitro methods.
Website: www.pkderm.com
YouTube chanel: PKDerm
Company page LinkedIn: PKDerm
Contact person via LinkedIn: Hanan Osman-Ponchet
PrediTox
PrediTox is an innovative French company that is committed to supporting its customers in evaluating the genotoxic and potentially carcinogenic properties of their samples or products. With more than 10 years of experience in this field, the PrediTox team knows how to meet the expectations of these customers thanks to its innovative techniques and personalized studies, with the aim of providing quality information while respecting new regulations and by not using animals. Our goal is to act at an early stage of development, where the quantities of products, the time to obtain results and the price are optimized.
Website : preditox.fr
Contact person via LinkedIn : Laure Khoury
Primacyt Cell Culture Technology GmbH
Primacyt develops, manufactures and distributes in vitro biological test systems for product development and safety applications in human and animal health and environmental safety. We offer one of the world’s most comprehensive portfolios of liver related products obtained from human and animal species like birds, fresh and seawater fish, rodents, and farm animals. Our liver cells have been successfully used for 2D and 3D cultures and microfluidic organ-on-a-chip applications.
Our offering includes primary human and animal cells, liver subcellular fractions, ex vivo human and animal skin explants, blood cells, serum products, and consumables for applications in environmental toxicology, preclinical testing and drug development for human and animals. Products are manufactured in accordance with OECD test guidelines when applicable.
We are GLP certified since 2006 and offer contract research studies for drug development and medical device testing. We have a validated technology platform to assay drug transporter activities in stably transfected HEK293-cells and primary hepatocytes. Since 2013 Primacyt serves as a reference laboratory for the validation of alternatives to animal experiments for the European Union.
Website: primacyt.com
Company page LinkedIn: Primacyt Cell Culture Technology GmbH
Contact person via LinkedIn: Dieter Runge
WeChat: PRIMACYT
ProtoQSAR SL
ProtoQSAR is a company of computational chemistry, specialized in the development and optimization of active compounds in diverse areas, like pharmacy, cosmetic or functional food. Our computer models are also an efficient alternative that allows the replacement of animal tests for compliance with regulatory standards such as the REACH regulation of the European Union.
Our activity consists in the development and application of computational methods for the evaluation of physicochemical, biological and/or (eco)toxicological properties of chemicals, either of natural origin or synthesized. Our computer tools allow us to work in a “virtual” environment, which has the following advantages over traditional laboratory tests:
Fast results: our methods reduce the time needed with in vitro and in vivo tests, thanks to its easy and immediate applicability to thousands of chemical structures, something impossible to perform experimentally.
Saving of material and financial resources, avoiding the costs associated with experimentation, both personal and laboratory equipment.
Limitation of animal testing (3Rs), which have to be performed only as a last resort, when no other scientifically reliable ways to demonstrate the impact of chemicals on humans and/or the ecosystem are available.
Regulatory validity: we help to comply with European directives for the registration and labeling of compounds (REACH, CLP, cosmetics, phytosanitary, etc.) and standards such as those set out in the ICH guidelines on impurities of pharmaceutical products. European legislation not only authorizes computational techniques as valid legal alternatives, but also encourages and stimulates them by the undoubted advantages they represent.
We work on very different kind of projects, because our technology can be used in different areas such as pharmacy, veterinary, cosmetic, agrochemical, functional food, etc.
– Website: protoqsar.com
Company page LinkedIn: ProtoQSAR SL
REGENHU
REGENHU is a Swiss MedTech company developing, producing, and commercializing 3D-bioprinting platforms, with a particular interest in application fields such as tissue engineering, drug screening, and drug discovery.
3D-Bioprinting is a powerful tool for in vitro drug discovery and personalized medicine and thus fully replacing the need of animal models and donor tissue. It allows to create tissue models imitating the complexity and functionality of healthy and/or diseased tissues in vitro. With the combination of multiple cell populations and materials in the same process in an automated and repetitive way, it is possible to create reliable tissue models. These are then used to rapidly screen the effect and efficacy of novel active compounds in drug discovery and to exploit patients’ cells for personalized medicine, enabling customized and on-chip diagnostics and treatments.
As a pioneer in bioprinting, REGENHU launched its first instrument to the market in 2009. Since then, the company’s main goal is to create and develop bioprinting technologies that can positively impact many medical and pharmaceutical fields. Based in Fribourg, Switzerland, REGENHU is commercially active in Europe, North America, and Asia. REGENHU’s global customer portfolio comprises key players in the biotech, pharma, cosmetics, and MedTech industries, in addition to leading universities worldwide.
– Website: www.regenhu.com
SABEU
IN STOCK! cellQART® Cell Culture Inserts – Direct from the original manufacturer.
Meet us in person at ESTIV2022
SABEU is actively working to accelerate the in vivo to in vitro transition. We manufacture plastics and membranes for off-the-shelf and customized life sciences products – all Made in Germany. Consequently, we can delivery very fast and are flexible to put any insight or requirement coming from you into a new product. With our home brand of cellQART®, we make the highest quality Cell Culture Inserts available at affordable prices. We offer a wide portfolio of Cell Culture Inserts in diverse pore sizes, membrane optics, individually packed and pre-loaded in cellQART® Well Plates. We work with industry leading quality standards to ensure 100% membrane parameter consistency and reproducible cell culture results. We are beyond excited to be part of the 3Rs transformation. Let’s work together towards a more Human and Humane world! For more information, visit our website and contact us on www.cellQART.com! Follow us on Twitter, LinkedIn and YouTube.
Website links:
Company page Twitter: cellQUART
Contact persons via LinkedIn:
SenzaGen
SenzaGen aims to be a leader in in vitro science and testing, driving the transition from animal testing to methods better suited to reflect human biology. The Company provides high-performance, non-animal test methods and innovation and consulting services based on state-of-the-art technology.
Recently, SenzaGen’s GARD®skin assay was adopted by OECD as part of Test Guideline 442E for in vitro skin sensitization. The scientific data published in the TG confirms the method’s high performance and demonstrated applicability for “difficult-to-test” chemicals such as complex mixtures, indirectly acting haptens, lipophilic compounds and metals, bringing new opportunities to the field of in vitro skin sensitization testing.
Website links:
Company page LinkedIn: SenzaGen
STEMCELL Technologies
Driven by science and a passion for quality, STEMCELL delivers over 2500 specialized products and services to accelerate scientific discovery.
Our ethically sourced human primary cell portfolio includes cryopreserved products from peripheral blood, cord blood, and bone marrow. We are also the world-leading provider of high-quality cell culture media for generating and culturing pluripotent stem cell-derived and organoid models. Additionally, our in-house Contract Assay Services (CAS) team specializes in primary cell-based assays and works with you to design and perform your drug development studies, helping you get the data you need.
Learn more at booth #16 or at www.stemcell.com
Website links:
Company page LinkedIn: STEMCELL Technologies
Ten Bio
Ten Bio is an international research partner organisation founded in 2020, backed by a heritage of decades of laboratory research and development. Our scientific team has a wealth of experience in a wide range of dermatology and skin biology. With labs in Dundee, Scotland and Kannapolis, NC, USA, we can offer our specialized services to global clients and provide our ready-to-use TenSkin™ model to the North American and European Markets.
At Ten Bio we provide optimal human skin models for all ex vivo skin testing needs. By restoring and maintaining physiological skin tension of real human skin, Ten Bio has succeeded in creating a truly functional and realistic ex vivo testing platform for skin-based research. Our technology and research services generate exacting and clinically relevant data to more accurately predict product safety and efficacy in vivo, thus lowering R&D costs and accelerating product development.
Our clients and research partners receive a wealth of support from their earliest engagement and can take advantage of our full spectrum of expertise, including experimental design and execution, as well as an extensive catalogue of SOP’s to accelerate R&D programs. We offer a comprehensive range of analysis techniques, including histological (including advanced microscopy), RT-qPCR, proteomics, transcriptomics and complete data analytics and interpretation packages for a wide range of application areas.
Website: www.ten-bio.com
Contact person via LinkedIn: Robyn Hickerson
TissUse
TissUse is a Berlin, Germany-based, biotechnology company, which has developed a unique “Multi-Organ-Chip” platform that provides unparalleled preclinical insight on a systemic level using human tissues. This enabling technology platform consists of a miniaturized construct that closely simulates the activity of multiple human organs in their true physiological context. TissUse’s Multi-Organ-Chips provide a new approach to predict, for example, toxicity, ADME profiles and efficacy in vitro, reducing and replacing laboratory animal testing and streamlining human clinical trials.
TissUse’s Multi-Organ-Chips have been utilized in a large variety of applications including drug development, cosmetics, food and nutrition and consumer products since 2012.
TissUse is additionally applying its platform and know-how to develop spin-off programs in a variety of tissue and organ repair areas starting with the cosmetic market of hair transplants.
Website link: www.tissuse.com
Company page LinkedIn: TissUse
Contact person via LinkedIn: Christine Schwenk
Company page Twitter: TissUse
Toxys
Toxys is a global leading expert that develops and offers a broad spectrum of unique animal-free laboratory tests for the safety assessment of novel medicines, chemicals, cosmetics and food. We are experts in the fields of genotoxicity, developmental toxicology and mechanistic toxicity testing. We develop assays with the focus on understanding the mode-of-action of toxic compounds. Our ToxTracker® assay accurately identifies genotoxic compounds, ReproTracker® detects developmental toxicity and the ToxProfiler™ assay provides a broad toxicity profile of compounds. Toxys is internationally recognised for its scientific expertise, high quality results and responsiveness.
Website links:
Company page LinkedIn: Toxys
Contact person via LinkedIn: Giel Hendriks
Universitat de Barcelona
Website: www.ub.edu
VITROCELL® Systems GmbH
VITROCELL® is specialised in the development of advanced in vitro exposure systems. VITROCELL® realizes turnkey installations for in vitro inhalation toxicology where gases, environmental atmospheres, nanoparticles and complex mixtures are analyzed on lung cells at the air/liquid interface.
The company offers relevant exposure technologies for continuous flow exposure, liquid aerosols using Cloud single droplet sedimentation as well as for small quantities of dry powders.
The product range includes systems for manual as well as for automated operation.
VITROCELL® in vitro Exposure Stations are designed for fully automated exposure of environmental pollutants to cells of the respiratory tract. This technology is well suited for long-term exposures > 24 hours.
VITROCELL® Skin modules and the VITROCELL® Skin Autosampler are specially designed for the exposure of tissue.
A complete set of dosimetry tools is an essential part of the product program.
The customers of VITROCELL Systems GmbH are leading research institutes, contract research organisations, regulatory authorities as well as the pharmaceutical and other industries throughout the world.
Website: www.vitrocell.com
Company page LinkedIn: VITROCELL® Systems GmbH
Watchfrog
Watchfrog Laboratory is exclusively dedicated to Endocrine Disruptors, testing chemicals and products.
From the inception of Laboratoire Watchfrog, our mission has been to provide a physiologically relevant diagnostic of whole organism endocrine activity while avoiding the use of laboratory animals.
The OECD (Organisation for Economic Co-operation and Development) has now published two test guidelines developed by Watchfrog based on aquatic embryos to contribute replacing the use of laboratory animals:
- The OECD Test Guideline n°248 aka XETA assay (Xenopus Eleuthero-embryonic Thyroid Assay);
- the OECD Test Guideline n°251 aka RADAR assay (Rapid Androgen Disruption Activity Reporter).
The publication by the OECD of this Test Guideline is a significant step in strengthening the available tools for applying European criteria for identifying Endocrine Disruptors, contributing to replace tests on mammals and reduce animal suffering,
The use of XETA in regulatory procedures and its acceptance by authorities paved the way for these embryonic alternative methods to provide a significant reduction in the cost to industry when performing chemical risk assessment.
Watchfrog Laboratory also provides tailored-made protocols to contribute to the endocrine safety of products, solid materials, fabrics, packaging…
For cosmetic ingredients and formulas, as OECD level 3 methods, XETA and RADAR provide the highest weight of evidence which is accessible under cosmetic regulation for a comprehensive assessment of endocrine mechanisms.
Website links:
Contact person via LinkedIn: Gregory Lemkine